 When atoms come together in chemical bonding, they share electrons. In chemistry, polarity refers to the way in which atoms bond with each other. Non-polar amino acids are a class of amino acids in which the variable R-group is comprised of mostly hydrocarbons the amino acids cysteine and methionine also feature a sulphur atom, but (due to its similar negativity to carbon) this does not confer any polar properties to either of these amino acids.Īmino acids are ordered from the most hydrophobic one, Isoleucine (I, on the left hand side) to the most hydrophilic one, Arginine (R, on the right hand side), according to the Kyte-Doolitle scale. Polarity of the amino acids affects the overall structure of a protein. 
All polar amino acids have either an OH or NH2 group (when in aqueous environment), and can therefore make hydrogen bonds with other suitable groups. The properties of the amino acid are due to the properties of the side chain or R-group. If no partial charges, it’s a nonpolar amino acid. While this won’t tell you if a given atom achieves a full charge, or only a partial charge it’s sufficient to let you know if there are atoms in a side chain with partial charges… Which means that side chain has polar elements. 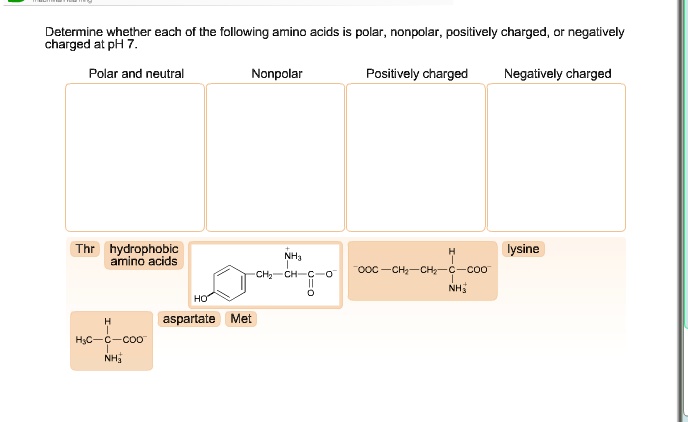
How do you know if an amino acid is polar or nonpolar? 9 How do you memorize polar and nonpolar amino acids?.6 What type of amino acid is asparagine?.5 Is glycine polar or nonpolar amino acid?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
